

Gene-edited islet cells survived 12 weeks in a type 1 diabetes patient with zero immunosuppression drugs, using CRISPR-Cas12b to achieve 100% HLA-II depletion.
Gene-edited islet cells survived 12 weeks in a type 1 diabetes patient without any immunosuppressive drugs. In this first-in-human study, researchers used CRISPR-Cas12b technology to create hypoimmune islet cells that achieved 100% HLA class II depletion and 85.8% HLA class I depletion. This matters because eliminating the need for immunosuppression could revolutionize transplant medicine by avoiding the serious side effects of lifelong immune-suppressing medications.
Let me break this down. For over a century, we've treated type 1 diabetes with insulin injections—a treatment, not a cure. When doctors transplant donor islet cells (the insulin-producing cells from the pancreas), patients typically need powerful immunosuppressive drugs for life to prevent rejection. These medications cause significant health problems and can be deadly. But what if we could edit the cells themselves to become invisible to the immune system?
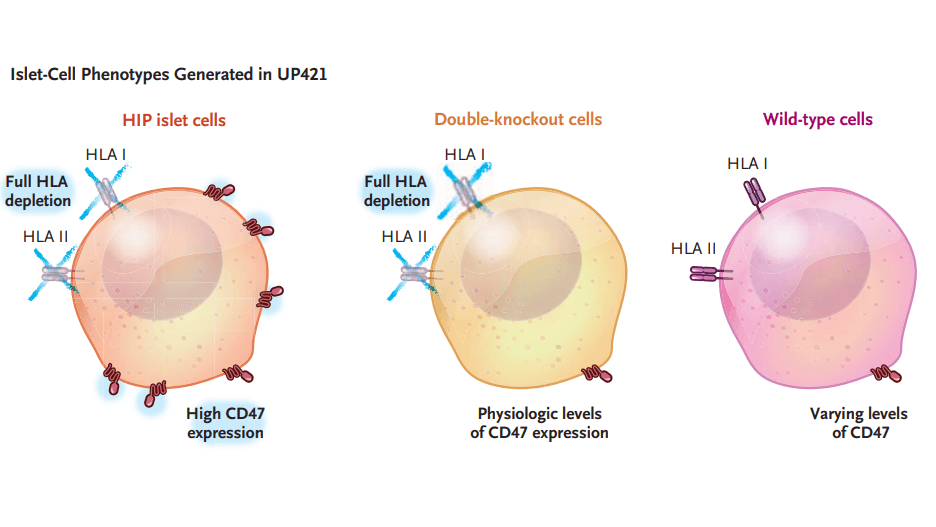
The research team used a sophisticated two-step editing process to create what they call "hypoimmune platform" (HIP) islet cells. First, they used CRISPR-Cas12b technology to knock out two critical genes: B2M (which controls HLA class I molecules) and CIITA (which regulates HLA class II molecules). Think of HLA molecules as identification badges that tell the immune system "this is foreign tissue—attack it." By removing these badges, the cells became harder for T-cells to recognize.
Here's the fascinating part: removing HLA molecules solved one problem but created another. Cells without HLA become targets for natural killer cells and macrophages through "missing-self recognition"—basically, the immune system saying "if you don't have proper ID, you're suspicious." So the researchers added a second modification: they used a lentiviral vector to overexpress CD47, a protein that acts like a "don't eat me" signal to innate immune cells.
The final product contained 85.8% HLA class I-negative cells, 100% HLA class II-negative cells, and 46.4% with high CD47 expression. The editing process didn't change the islet composition—about 66% remained functional beta cells capable of producing insulin.
The 42-year-old participant, who had lived with type 1 diabetes for 37 years, received 79.6 million gene-edited islet cells injected into his forearm muscle. He took zero immunosuppressive medications—a radical departure from standard transplant protocols.
The results were striking. While the unedited "wild-type" islet cells in the transplant triggered massive immune responses—with T-cell activation peaking at day 7 and antibody responses developing between days 14 and 21—the fully edited HIP islet cells showed no detectable immune response throughout the entire 12-week study period. The participant's immune system essentially ignored the gene-edited cells while attacking the unmodified ones.
C-peptide measurements (a marker of insulin production) remained stable from 7 days through 12 weeks, and the cells showed glucose-responsive insulin secretion during meal tolerance tests. Before transplantation, the participant had undetectable C-peptide levels; after transplantation, his levels increased in response to meals at 4, 8, and 12 weeks. His glycated hemoglobin level dropped by approximately 42% over the follow-up period.
PET-MRI imaging at 12 weeks confirmed the graft's survival and vascularization, showing specific radiotracer uptake in the transplanted areas—evidence that the cells were alive, functional, and receiving blood supply.
The evidence suggests this technology could eliminate the need for lifelong immunosuppression in transplant recipients. Building on earlier CRISPR applications in cell therapy, particularly CAR T-cell treatments, this approach demonstrates that gene editing can create "universal donor" cells that evade immune rejection.
However, let's acknowledge the limitations explicitly stated in this study. The dose of 79.6 million HIP islet cells was only 7.1% of the amount typically needed to achieve insulin independence (approximately 11,547 islet equivalents per kilogram of body weight). The researchers intentionally used this low dose to meet safety requirements for a first-in-human trial. The participant still required insulin injections, though his glycemic control improved.
The 12-week follow-up period is relatively short—previous studies in non-human primates showed stable function for 6 months, but long-term human data doesn't exist yet. The product contained some unedited cells that did trigger immune responses, potentially sensitizing the patient to future transplants. Four adverse events occurred (mild thrombophlebitis and arm paresthesia), though none were serious or directly related to the gene-edited cells.
The study enrolled only one participant, making it impossible to draw broad conclusions. Questions remain about whether repeated transplants would maintain effectiveness, whether the editing increases cancer risk through insertional mutagenesis (though CAR T-cell data from 783 patients suggests this risk is minimal), and whether full-dose transplants would actually achieve insulin independence.
Still, this proof-of-concept study represents a potential paradigm shift. If scaled up, hypoimmune islet cells could offer a functional cure for type 1 diabetes without the devastating side effects of immunosuppression—transforming transplant medicine from managing rejection to engineering acceptance.
Yes. In this 2025 first-in-human study, CRISPR-Cas12b edited islet cells survived 12 weeks in a type 1 diabetes patient with zero immunosuppressive drugs. The fully edited HIP cells showed no detectable immune response while achieving 100% HLA class II depletion and 85.8% HLA class I depletion.
Researchers used CRISPR-Cas12b to knock out B2M and CIITA genes (removing HLA class I and II molecules that trigger rejection), then used lentiviral transduction to overexpress CD47 protein. This two-step process created hypoimmune islet cells that evade both adaptive and innate immune responses. They transplanted 79.6 million edited cells and monitored for 12 weeks.
The current study used only 7.1% of the dose needed for insulin independence (79.6 million cells vs ~1.1 billion needed), enrolled only one participant, had a 12-week follow-up period, and the transplant contained some unedited cells that could sensitize patients to future transplants. Long-term safety beyond 12 weeks remains unknown.
This article has been reviewed by a PhD-qualified expert to ensure scientific accuracy. While AI assists in making complex research accessible, all content is verified for factual correctness before publication.
CRISPR-Cas9 Allele-Specific Trisomy 21 Chromosome Elimination in Human iPS Cells
A 2025 study used allele-specific CRISPR-Cas9 to eliminate the extra chromosome 21 from Down syndrome iPS cells, achieving a 30.6% karyotype correction rate in clones, outperforming non-specific methods and restoring gene expression linked to nervous system development.
Why Your Body Fights to Regain Lost Weight
Turns out, fat cells remember obesity in their DNA. Here's what that means for weight loss.
Your Brain’s Nightly Deep Cleaning
Sleep isn’t just for dreams—your brain is busy taking out the trash.
No comments yet. Be the first to share your thoughts!
Get notified when we publish new articles. No spam, unsubscribe anytime.